
Our company's is one of world’s few integrated OEM company of needles. We manufacture everything from plastic molded items such as hubs and caps to cannulas, and carry out all assembly, packaging, and sterilization in-house. The greatest merits of our integrated manufacturing system are our comprehensive quality management system, and our ability to provide the customer with reliable products.
We have strict inspection procedures in all processes and are certified under international standards for quality assurance of medical equipment such as ISO 13485, CE Mark, and the FDA. Our unique technology accumulated over many years is utilized in the manufacture of fine tube, and polishing and coating of needle tips, and is highly evaluated as ‘Japanese Quality’.
 |
Tube Manufacture A process for the manufacture of fine tube for injection needles from stainless steel sheet. The stainless steel sheet used is a special product with hardness and tenacity optimized for fabrication of injection needles. The manufacturing process involves rolling stainless steel sheet and welded into fine tube. In the floating process, the diameter and wall thickness are repeatedly reduced to the approximate dimensions required for the injection needles. |
|
 |
||
 |
Fabrication into stainless steel tube The stainless steel tube from the tube manufacturing process is annealed to a hardness suitable for fabrication. Diameter and wall thickness are then adjusted to specification in the drawing process, the tube is passed through a straightening machine to produce a straight tube, which is then cut to length. Stainless steel tube is sampled at a rate of 6 in 5000. Five of the samples are inspected, and one retained in storage. |
|
 |
||
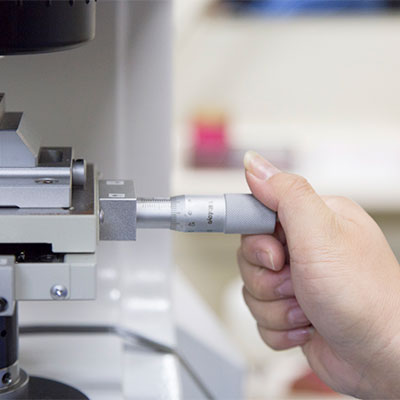 |
Tube Inspection The fine stainless steel cut tube for use in injection needles is sent to the inspection room and inspected by veteran staff for kinks, scratches, and contamination etc. Diameter, wall thickness, and deflection etc. are accurately measured in sampling inspections, and further inspections conducted to evaluate breaking resistance, bending strength, elasticity, appearance, and inside diameter. Testing is conducted in accordance with an inspection plan, and results recorded. |
|
 |
||
 |
Fabrication Into Injection Needles Stainless steel tube manufactured to the thickness of the injection needle is further processed according to specified application. The needle tip is ground with a high-speed grinding wheel. Burrs on the tips of the smooth cannula of cleaning needles etc. are removed by an electrolytic process, surface-treated, cleaned down to the base stainless steel surface, and sent to a clean room. The needle tips are fabricated to various requirements, and the manufacturing process is constantly reviewed according to a wide range of requirements. Our company accepts the challenge of developing new injection needles in response to new medical techniques constantly in development around the world. |
|
 |
||
 |
Assembly All assembly work is carried out in the clean room. All finally cleaned cannulas are inspected and sent to the assembly process. The cannula and hub are glued together in the assembly process, coated, and the cap attached. All processes are automated, and products without cannula and defective needles, as well as points (tip bend), are inspected for all needles. Assembled injection needles are inspected for adhesion strength and puncture resistance. Injection needles with low resistance to penetration resistance, and consequently less pain, are highly desirable in many countries, and are available as cannula and bulk products to suit customer requirements. Sampling inspection: Adhesive strength, puncture resistance, needle bending, appearance |
|
 |
||
 |
Packaging and Sterilization The assembled injection needles are packed 10 per sheet, each sheet inspected, and packed 10 sheets per box. Each box is fed to the sterilizing process and shipped as an OEM product. Since Our company's manufactures OEM products for many countries, all packaging and boxes are printed with individual customer branding. The final processes of packaging, boxing and sterilization can be implemented in accordance with the requirements of each OEM customer. It is therefore possible to specify sterilized, un-sterilized, boxless etc. formats as necessary. We also offer a sterilization-only service. Packaging Boxing Sterilization |
|
 |
 |
 |
 |
 |
 |